Abstract
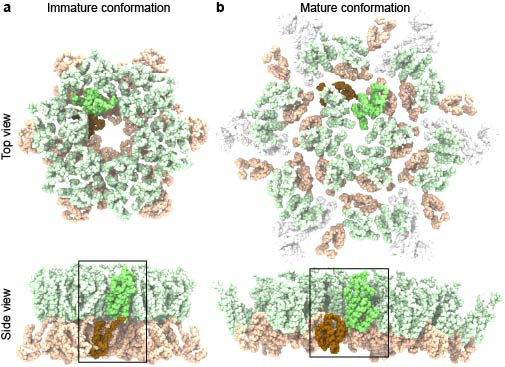
During HIV-1 maturation, capsid proteins (CA) are proteolytically cleaved from the immature Gag lattice and undergo conformational transitions in which the amino-terminal and carboxy-terminal domains rearrange into mature conformations prior to assembling into fullerene complexes. Little is known, however, of the structural transitions and energetic principles governing this transformation. Here, we reveal the dynamical mechanisms of CA maturation using all-atom and enhanced sampling simulations. Free energy calculations demonstrate that immature CA is intrinsically unstable as a monomer and rapidly converts into a mature form that is consistent with x-ray crystallographic and cryo-electron microscopy structures. Analysis of the minimum free energy pathway indicates that conformational change in CA proceeds through stepwise, ratcheting transitions, and we identify a metastable intermediate distinct from the immature and mature endpoints. Energy decomposition analysis uncovers the interdomain residue-residue interactions, particularly charged contacts, that stabilize the mature state. These results reveal the interactions and molecular mechanisms that drive the conformational changes required for HIV-1 capsid domain maturation.